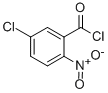
Benzoyl chloride, 5-chloro-2-nitro-
5-CHLORO-2-NITRO-BENZOYL CHLORIDE
CAS: 41994-44-9
Molecular Formula: C7H3Cl2NO3
Benzoyl chloride, 5-chloro-2-nitro- - Names and Identifiers
| Name | 5-CHLORO-2-NITRO-BENZOYL CHLORIDE |
| Synonyms | 5-CHLORO-2-NITRO-BENZOYL CHLORIDE Benzoyl chloride, 5-chloro-2-nitro- |
| CAS | 41994-44-9 |
Benzoyl chloride, 5-chloro-2-nitro- - Physico-chemical Properties
| Molecular Formula | C7H3Cl2NO3 |
| Molar Mass | 220.00962 |
| Storage Condition | Room Temprature |
Benzoyl chloride, 5-chloro-2-nitro- - Upstream Downstream Industry
| Downstream Products | 1-(5-CHLORO-2-NITROPHENYL)-1-ETHANONE Methyl 5-chloro-2-nitrobenzoate |
Benzoyl chloride, 5-chloro-2-nitro- - Introduction
CHLORIDE, molecular formula C7H3Cl2NO3, molecular weight 215.01. The following is a description of the properties, uses, preparation and safety information of the compound:
Nature:
-Appearance: Colorless to light yellow crystal or crystalline powder
-Melting point: 81-83 ° C
-Boiling point: 220-222 ° C
-Density: 1.61 g/mL
-Solubility: Slightly soluble in water, soluble in organic solvents such as ether, chloroform, dichloromethane, etc.
Use:
-CHLORIDE is an important organic synthesis intermediate, commonly used in the preparation of drugs, pesticides and dyes and other compounds.
-It can be used as an acylation reagent for the synthesis of other organic compounds, such as acylated derivatives, amino acid derivatives, etc.
-The compound can also be used for the preparation of drugs for the prevention and treatment of tuberculosis, anticancer drugs, etc.
Method:
-the preparation of CHLORIDE can be obtained by reacting phthalic anhydride with nitric acid. First, phthalic anhydride and nitric acid are reacted with sulfuric acid to form nitrobenzoic acid. The resulting nitrobenzoic acid is then reacted with thionyl chloride to further produce 2-nitro-5-chlorobenzoic acid. Finally, 2-nitro-5-chlorobenzoic acid was converted to CHLORIDE by reaction with thionyl CHLORIDE.
Safety Information:
-CHLORIDE is an organochlorine compound that is irritating and corrosive. Exposure to the substance may cause irritation of the skin, eyes and respiratory tract, and even severe burns.
-Wear appropriate personal protective equipment such as goggles, gloves and respiratory protection when using the compound.
-Avoid inhalation, skin contact and eye contact when handling the substance.
-Store away from fire and oxidizing agents, and store in a closed container.
-It is recommended to use in a well-ventilated laboratory environment and comply with relevant safety procedures. If necessary, professional guidance should be sought.
Nature:
-Appearance: Colorless to light yellow crystal or crystalline powder
-Melting point: 81-83 ° C
-Boiling point: 220-222 ° C
-Density: 1.61 g/mL
-Solubility: Slightly soluble in water, soluble in organic solvents such as ether, chloroform, dichloromethane, etc.
Use:
-CHLORIDE is an important organic synthesis intermediate, commonly used in the preparation of drugs, pesticides and dyes and other compounds.
-It can be used as an acylation reagent for the synthesis of other organic compounds, such as acylated derivatives, amino acid derivatives, etc.
-The compound can also be used for the preparation of drugs for the prevention and treatment of tuberculosis, anticancer drugs, etc.
Method:
-the preparation of CHLORIDE can be obtained by reacting phthalic anhydride with nitric acid. First, phthalic anhydride and nitric acid are reacted with sulfuric acid to form nitrobenzoic acid. The resulting nitrobenzoic acid is then reacted with thionyl chloride to further produce 2-nitro-5-chlorobenzoic acid. Finally, 2-nitro-5-chlorobenzoic acid was converted to CHLORIDE by reaction with thionyl CHLORIDE.
Safety Information:
-CHLORIDE is an organochlorine compound that is irritating and corrosive. Exposure to the substance may cause irritation of the skin, eyes and respiratory tract, and even severe burns.
-Wear appropriate personal protective equipment such as goggles, gloves and respiratory protection when using the compound.
-Avoid inhalation, skin contact and eye contact when handling the substance.
-Store away from fire and oxidizing agents, and store in a closed container.
-It is recommended to use in a well-ventilated laboratory environment and comply with relevant safety procedures. If necessary, professional guidance should be sought.
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: 5-CHLORO-2-NITRO-BENZOYL CHLORIDE Request for quotation
CAS: 41994-44-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41994-44-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 5-CHLORO-2-NITRO-BENZOYL CHLORIDE Visit Supplier Webpage Request for quotation
CAS: 41994-44-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 41994-44-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 5-CHLORO-2-NITRO-BENZOYL CHLORIDE Request for quotation
CAS: 41994-44-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41994-44-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 5-CHLORO-2-NITRO-BENZOYL CHLORIDE Visit Supplier Webpage Request for quotation
CAS: 41994-44-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 41994-44-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Benzoyl chloride, 5-chloro-2-nitro-
Cyclopropanepropanamine, 1-methyl-
95470-68-1
4-Pyridinecarboxaldehyde, 5-bromo-2-methoxy-
3-丁烯-1-基氨基甲酸酯
DL-3-Amino-3-(2-methylphenyl)propanoic acid
(S)-TERT-LEUCINE METHYL ESTER
4-(DIMETHYAMINO)CINNAMICACID
Diethyl 2-((pyridin-3-ylamino)methylene)malonate
2-氯-4-三氟甲基-1,3-噻唑-5-羧酸苄酯
Cyclopropanepropanamine, 1-methyl-
95470-68-1
4-Pyridinecarboxaldehyde, 5-bromo-2-methoxy-
3-丁烯-1-基氨基甲酸酯
DL-3-Amino-3-(2-methylphenyl)propanoic acid
(S)-TERT-LEUCINE METHYL ESTER
4-(DIMETHYAMINO)CINNAMICACID
Diethyl 2-((pyridin-3-ylamino)methylene)malonate
2-氯-4-三氟甲基-1,3-噻唑-5-羧酸苄酯
Downstream Products for Benzoyl chloride, 5-chloro-2-nitro-

